No.
Ingredient name
INCI name
CAS number
Details
No data
USA-The FDA Monograph Ingredients (OTC) (Sunscreen active ingredients)
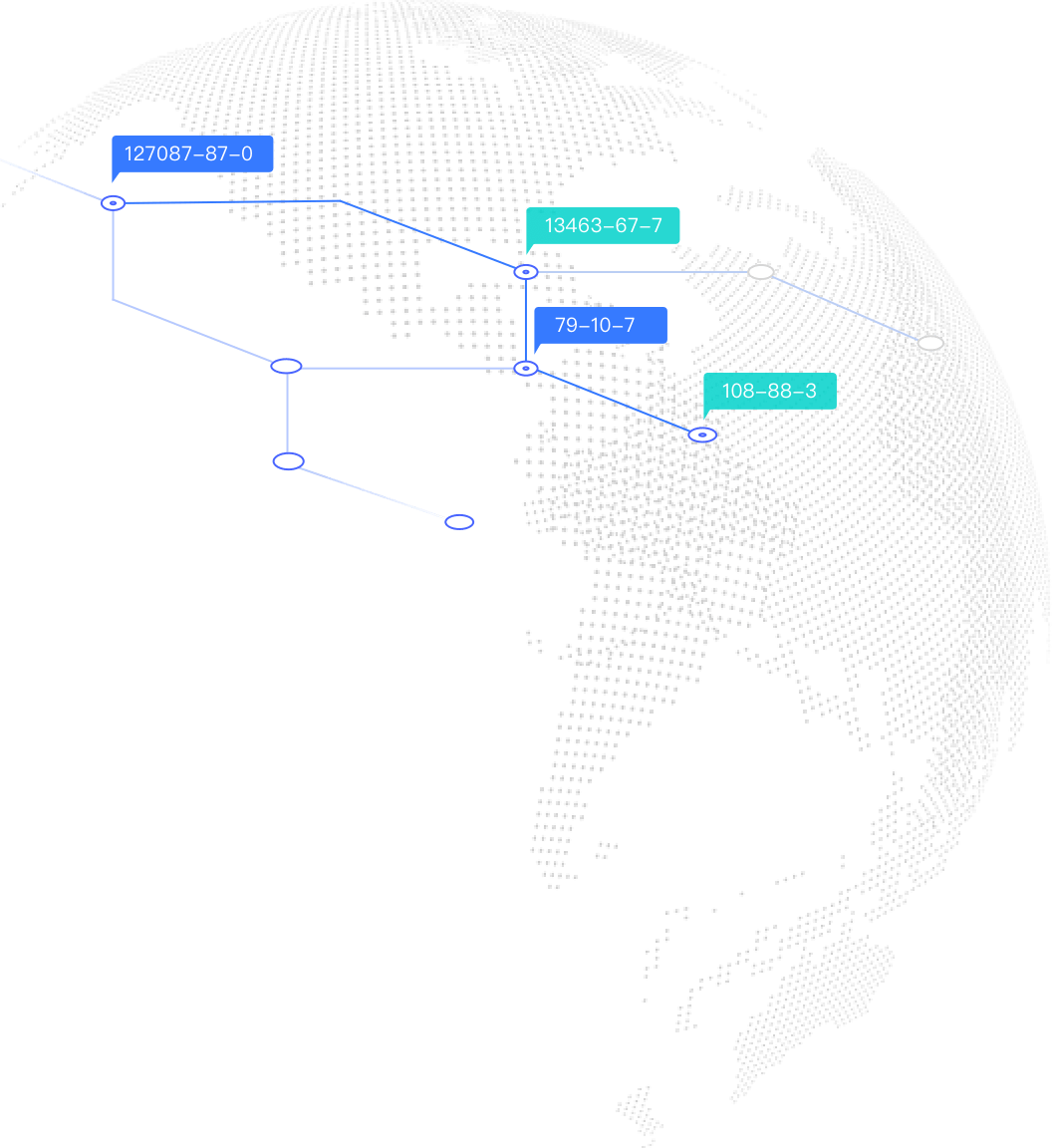
The Sunscreen Drug Products for Over-the-Counter Human Use is included in the FDA OTC Monographs which outlines the active ingredients that are generally recognized as safe and effective for use in OTC sunscreen products.
For more information, please contactchemicals@cirs-group.com

